Internal audit checklist iso 134851/30/2024  
Reporting and Compliance: Visure Requirements offers reporting capabilities that allow you to generate custom reports and metrics to monitor project progress, traceability coverage, risk status, and compliance with ISO 13485.This ensures that any changes made to the medical device development are properly evaluated, approved, and documented, aligning with ISO 13485 requirements. The platform supports change request tracking, impact analysis, and change control processes. Change Management: With Visure Requirements, you can effectively manage changes to requirements, design, and other project artifacts.This enables you to demonstrate traceability from user needs to design, verification, and validation, as required by ISO 13485. Traceability: Visure Requirements facilitates end-to-end traceability, allowing you to establish and maintain traceability links between requirements, design elements, tests, and other artifacts.This helps in meeting ISO 13485 requirements related to document control and record keeping. You can create, review, and approve documents, ensuring version control and maintaining a centralized repository of controlled documents. Document Control: The platform provides robust document control capabilities, enabling you to manage documents and records related to your quality management system.You can define risk criteria, perform risk assessments, and track risk mitigation activities, ensuring compliance with ISO 13485 requirements. Visure Requirements offers built-in risk management features, allowing you to identify, assess, and mitigate risks associated with your medical device development. Risk Management: ISO 13485 places a strong emphasis on risk management.You can track changes, perform impact analysis, and ensure that all requirements are adequately addressed. It allows you to define requirements, link them to applicable regulations and standards, and ensure traceability throughout development. Requirements Management: Visure Requirements enables you to capture, organize, and manage requirements in a structured manner.Here are some key capabilities of Visure Requirements that are relevant to ISO 13485: The platform’s traceability management capabilities allow for establishing and maintaining traceability links between requirements, design elements, tests, and other artifacts, ensuring compliance with regulatory standards. 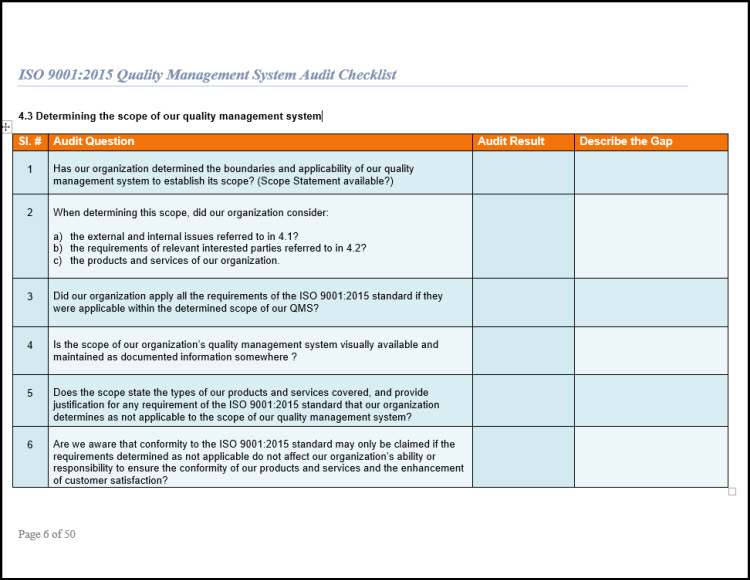
It facilitates efficient issue and defect tracking, ensuring that problems are captured, managed, and resolved effectively. Visure Requirements excels in providing end-to-end support for risk management, enabling users to identify, evaluate, and mitigate risks associated with medical device development. These esteemed clients bear testimony to Visure’s ability to meet their demanding needs and contribute to their success. Renowned organizations in the medical device sector, including Glaxo Smith Kline (GSK), Solta Medical, and Health Net, have placed their trust in Visure as their requirements management solution. The platform also extends its capabilities to quality analysis, requirements versioning, and robust reporting. It offers comprehensive integration throughout the entire Application Lifecycle Management (ALM) process, encompassing essential areas such as risk management, issue and defect tracking, traceability management, change management, and more. Visure Requirements is a widely recognized and trusted requirements management tool, particularly in the medical device industry. Best ISO-13485 Compliance Tools Visure Requirements ALM Platform In this article, we will discuss some great requirements management software organizations can use for ISO-13485 compliance, standard checklist, and essential templates. Compliance with this standard demonstrates a company’s commitment to producing safe and effective medical devices while maintaining a focus on quality and customer satisfaction. The purpose of ISO 13485 is to ensure that medical device manufacturers establish and maintain an effective quality management system to consistently meet customer and regulatory requirements. It outlines the requirements for organizations involved in the design, development, production, installation, and servicing of medical devices. 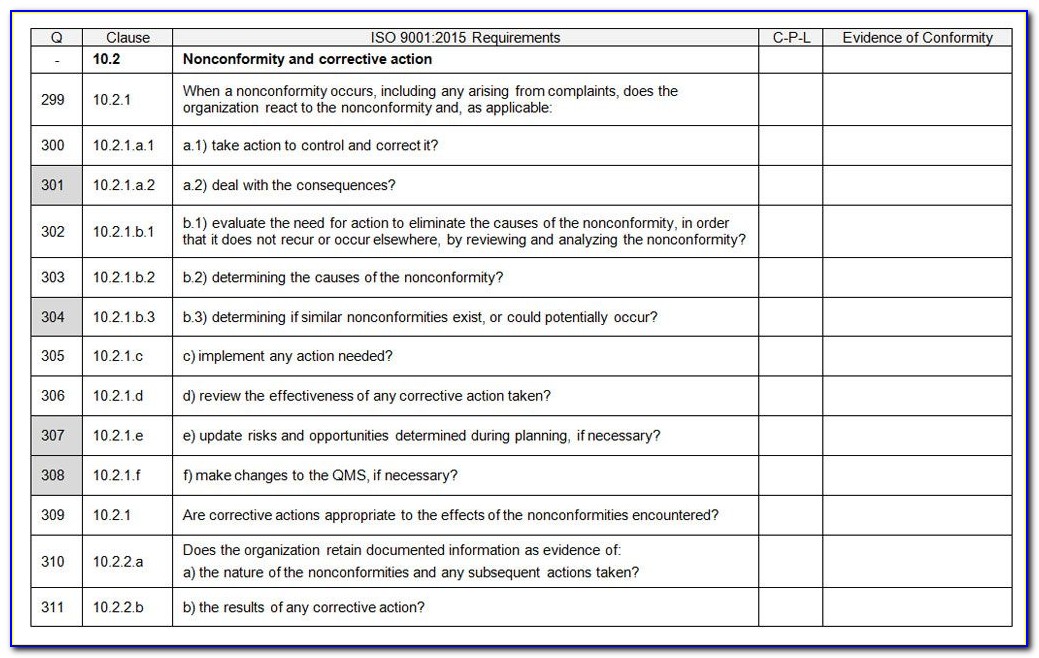
ISO 13485 is an international standard for quality management systems (QMS) specifically designed for the medical device industry. Requirements Management & Traceability with Microsoft Office Word and Excel.Optimizing for DO-178C & DO-254 Certification.Leveraging AI to Accelerate Requirements Management Process.Requirements AI- Course Online Training.Model Based Systems Engineering (MBSE) Online Training Course. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
